LEARNING OBJECTIVES
- ICH GCP and ICH GCP R3 Addendum
- EU Clinical Trials Regulation (EU CTR)
- Dutch legislation: Medical Research Involving Human Subjects Act (WMO), WGBO and Wet BIG
- Roles in clinical research
- Privacy in clinical research (GDPR)
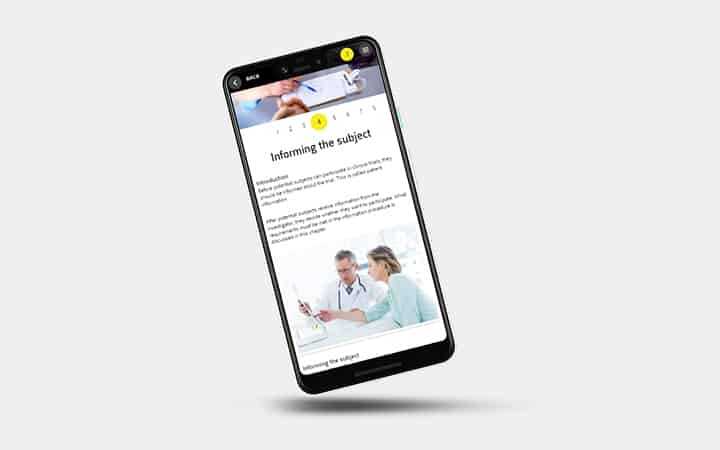
- Informing research participants
- Essential records
- Review by the Ethics Committee
- Safety reports and safety of your participants, incl. AEs, SAEs, and SUSARs
- Quality management
- Amendments and derogations
- Final report
- Audits and inspections
DESCRIPTION
The expert course to GCP in the Netherlands is the self-paced online training solution for researchers, pharma professionals, and research staff who conduct clinical trials in the Netherlands, and need to be aware of the Good Clinical Practice guidelines, EU laws, and the Medical Research Involving Human Subjects Act (WMO) applicable to clinical research with human participants. It is the best choice for those who like the flexibility of online learning at their own pace and time. This course translates the regulatory framework to practice and gives you the tools to directly implement the knowledge in your daily practice.
- Transcelerate acknowledged WMO/GCP certificate
- Efficient and interactive learning
- Easy accessible via smartphone, tablet, or desktop
- Self-paced and modular
- Accredited by PE-online quality registers
The course consists of 7 online training modules in a logical order: from the design to close-out of a clinical trial. It represents a total workload of 10-12 hours. The modules are divided into chapters of 10-30 minutes and consist of the guidelines which apply to international clinical trials, including the Declaration of Helsinki, ICH GCP E6 Addendum R3, U Clinical Trials Regulation (EU CTR), the EU Privacy law, Medical Research Involving Human Subjects Act (WMO), WGBO and Wet BIG.
After you have completed all components of the GCP e-learning course for clinical trials in the Netherlands you will receive a WMO/GCP Certificate, recognized by Transcelerate and accredited by many quality registers.
All training courses include one-year access to the online training. With our continuous learning model, we will keep you up-to-date with changing regulations and guidelines.
Enroll now to get started with the online learning modules on your smartphone, tablet, or desktop computer.
COURSE CONTENTS
Module: Introduction
- Introduction to medical research
- Types of clinical research (interventional, therapeutic, multi/single center)
- History of legislation and regulations clinical research (Code of Nuremberg, Declaration of Helsinki)
- The principles of ICH GCP incl. R3 Addendum
- Laws and regulation clinical research Europe (EU directives 2001 and 2005, EU Regulation 536/2014)
- Legal framework for clinical research in the Netherlands (WMO, WGBO, Wet BIG, AVG) and scope
- The roles in clinical research: sponsor, METC, competent authority CCMO or Ministry of Health (VWS), monitor, auditor, researcher, research professional
- Additional requirements for clinical trials of a medicinal product
- Phases of a clinical trial of a medicinal product
Module: Design
- Protocol development and content
- Select research team
- Selection of investigators and research locations
- Selection criteria for researchers and research locations
- Create and store essential documents (Investigator Site File)
- Risk inventory and assessment
- Setting up quality assurance: monitoring plan, auditing, DSMB, SOPs
Module: Preparation
- Package, label, import and deliver a research product
- Drafting product information (Investigators Brochure, IMPD)
- Drafting patient information and informed consent with Model Subject Information 2019
- Contracts and agreements
- Privacy legislation (AVG)
- Insurance (participant insurance and liability)
Module: VGO
- The process
- Visualization of the Local Feasibility procedure
- For which types of research does the VGO apply?
- The 4 steps of the Local Feasibility procedure
- Part A and Part B of the VGO
- Relationship between VGO and other documents
- The timelines of the Local Feasibility procedure
Module: Submission
- Composition of standard research file
- Composition and procedure of the review committee (METC or CCMO)
- Review by an ethical review committee
- Review by the competent authority: CCMO or Ministry of Health (VWS)
- Review timelines and submission of amendments
- Review process and approval
- Conditions and obligations after approval
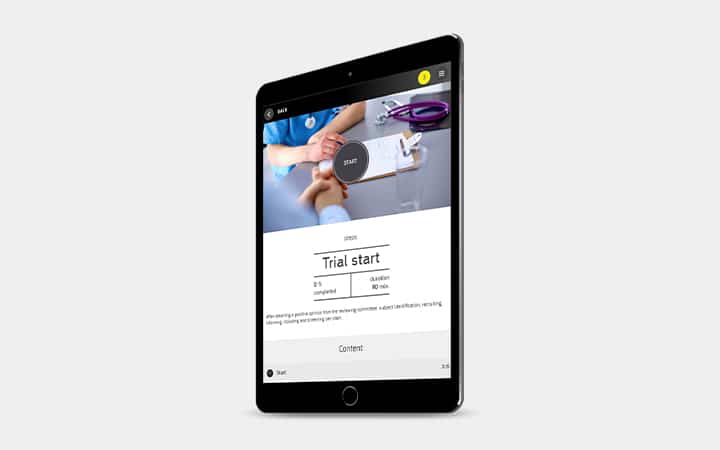
Module: Start
- Delegate tasks and Initiation Visit
- Supply, storage, and use of the investigational product in a trial
- Recruitment of participants
- Informing participants
- Informed Consent procedure
- Randomization and coding
- Privacy of data (AVG)
- Requirements for research with vulnerable participants
Module: Conduct
- Amendments, deviations, and changes in protocol and trial
- Adding new research sites and investigators
- Safety reports (AE / SAE / SUSAR)
- Safety participants medical care and DSMB
- Documentation and data management (source, CRF, database)
- Monitoring/ Auditing/ Inspection
- Quality assurance and risk management (R3 Addendum)
- Progress reports
Module: Archive (Close-out)
- Regular completion of a trial
- Preliminary closing of a trial
- Report end of the trial
- Requirements for Clinical Study Report
- Storage and archiving trial documentation
- Retention deadlines for trial documentation
Module: Knowledge Test
- 40 questions
- Available in English and Dutch
- Feedback per question (afterwards)
- 3 attempts
- Pass with a score of 80 or higher







matyas@igina.net –
Amazing training that I use every day in my work!